Introduction
What is a pest?
An agricultural pest is any organism or infectious agent that causes stress or damage to a desired plant or plant product. For example, a weed is a pest if it competes with a crop for resources, causing the crop stress it otherwise would not have. Infectious bacteria, fungi, and viruses cause diseases and are therefore pests. These small pests are transferred from one plant to another through water, through the air, by insects, or by larger animals (including humans). The most familiar pests are insects, ranging in size from the small whitefly to large swarms of locusts. Larger pests such as birds, mice, and rabbits can also cause damage in the field.
Some pests affect agricultural products in storage, such as cereal and pulse grains (Sallam, 1999; Manandhar et al., 2018). However, this article will focus on pest management in the field. In the remainder of this article, the term ‘pest’ will refer only to agricultural pests that affect crops in the field and will focus primarily on insect pests.
Globally, roughly 1 million insect species have been named. Scientists estimate that over 5.5 million more species exist but have yet to be discovered (Stork, 2018). The vast majority of insect species are beneficial or have little impact on crop production or human activity. However, a small portion (perhaps only 1% of insect species) are considered pests (Omkar, 2018). Currently, the most severe agricultural pests are non-native species that were introduced and spread through a region, for example through the import and export of agricultural products, in/on plant material, in soil, or through severe weather events. Natural predators that would otherwise keep their populations in balance may not be present outside of the insects’ native areas. Sometimes, native insects that did not previously impact farmers’ crops will switch host plants to new species that farmers start to grow; this can cause the insects to be elevated to pest status. However, please understand that the majority of insects do not damage crops. In fact, many are important parts of an integrated pest management plan because they help control outbreaks of other insect species.
Concerns in pest management
Pesticides have become a vital part of sustained food production to meet the needs of a growing global population. However, widespread pesticide use poses concerns to human health, the environment, and long-term sustainability. As an example, the process of pests becoming resistant to pesticides reduces the long-term effectiveness of those pesticides.
Climate change, the introduction of new pests, and several other factors have increased pest pressures in the tropics, where food insecurity is already a major concern. Pests contribute greatly to crop loss, and farmers in resource-limited regions experience some of the most significant effects (Chakraborty and Newton, 2011). For example, over the past three years in East Africa, the combined drought and fall armyworm (Spodoptera frugiperda) infestation resulted in maize crop losses of up to 100% for some farmers (FAO, 2017; Sisay et al., 2019).
Integrated Pest Management
The current global situation calls for a multi-pronged approach to pest management. To be widely applicable, this approach must provide farmers with options to control pests at various scales of production (from small farms to very large operations) with a diversity of resources. Integrated pest management (IPM), a strategy based on farmer innovations, is highly adaptable to specific contexts and reduces dependency on pesticides while still recognizing their use. IPM has gained traction since 1989; in particular, Farmer Field Schools (FFS) have effectively educated farmers about IPM (Peshin et al., 2009).
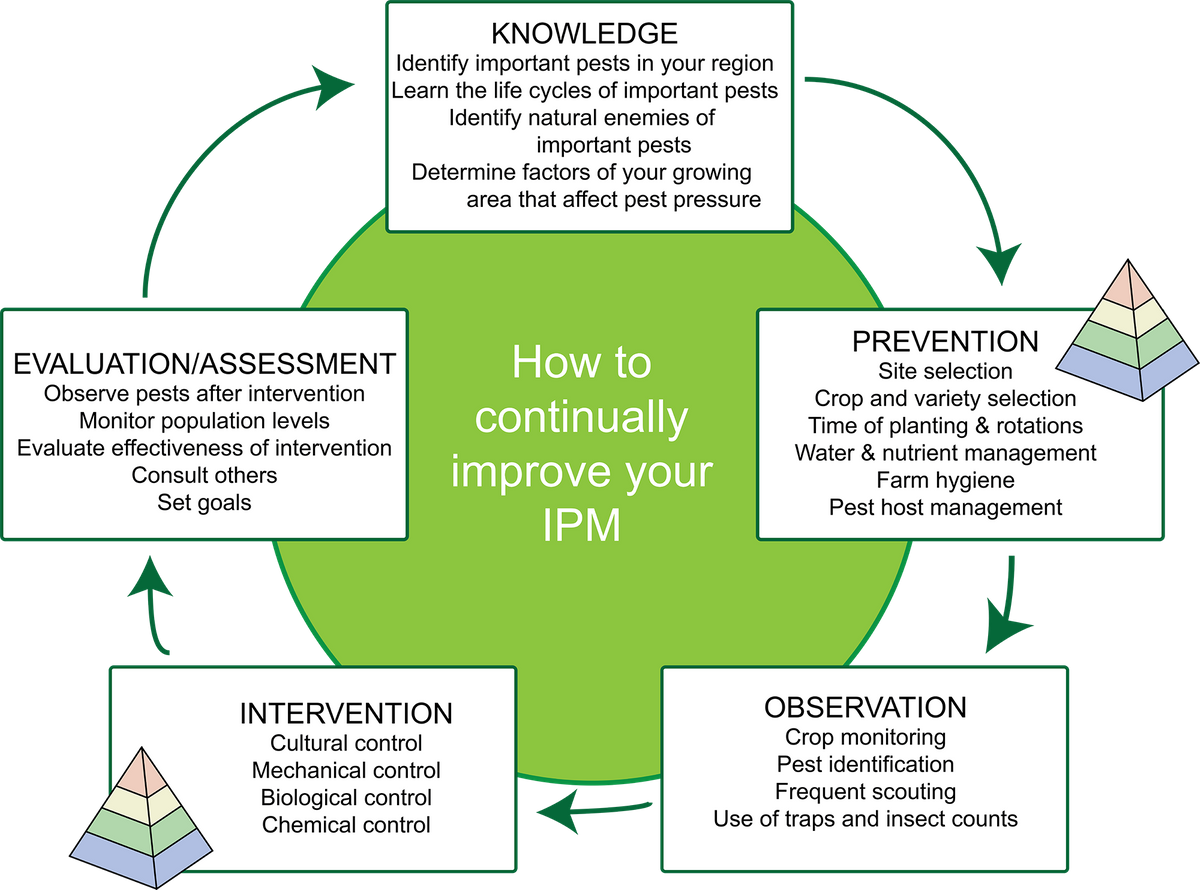
IPM is a management approach that focuses on the larger system and aims for long-term prevention of pests using a combination of preventative and suppressive control strategies. An individual farmer’s IPM plan should constantly improve as it cycles, as shown in figure 1. This article will address each stage in the cycle of improving your IPM plan starting with knowledge.
Figure 1. Stages of an example IPM cycle. Planning can start at any stage of the cycle, and the order of stages is flexible. The pyramid icon indicates strategies that prevent or suppress insect pests. Source: Adapted from farmbiosecurity, Creative Commons Attribution 3.0 license
Knowledge
Learning is useful at any time in a continually improving IPM strategy, but can be especially advantageous when beginning an IPM plan. Obtaining some of the important information may require community collaboration.
Identify important pests in the region
Seasonal insect pressure can change from year to year, due to the introduction of new pests from global trade and/or from the abiotic spread of pests and disease. It is important to record pests that farmers regularly see during different growing seasons; this will help you understand which pests are common, and when they tend to arrive each season.

Figure 2. Cowpea bruchid (Callasobruchus maculatus) eggs (left) and adults (right). On the cowpea seed, you can also see a hole where one of the larva emerged. Source: Tim Motis
Learn the life cycles of important pests
Pests’ lifespans and life cycles vary greatly. You should be able to identify common pests in many different life stages, such as egg, larva, nymph, pupa, or adult (Figure 2). If you can recognize stages in pests’ life cycles, you will be better able to intervene, because some interventions are only effective at certain life stages. Information such as lifespan and/or the presence of a winged stage allows you to estimate the potential rate of spread and/or mobility of the pest population.
Identify natural enemies of important pests

Figure 3. Ladybug (Harmonia sp.), an insect pest predator. The larva in the left photo is feeding on aphids. The right photo shows an adult ladybug. Both were on sorghum leaves at ECHO in Florida. Source: Tim Motis
Natural enemies of pests may live in the environment and already help control the pest population naturally. Some natural enemies are well-known (Figure 3), while others can be determined by carefully observing interactions between pests and other species. Once you know of natural enemies, you can record their relative abundance before and during the growing season.
Determine factors in the growing area that affect pest pressure
Pay attention to factors in the growing area that affect pest pressure. These may be environmental, such as trends observed in the field during and after the growing season. They may also include cultural, economic, political, and social dynamics. Here are some examples of questions to think about and discuss at a community level:
- Does everyone plant at the same time? Some pests only affect crops during specific windows of plant development. If all farmers in a region plant a crop at the same time, hypothetically they will all experience similar pest pressure. However, if one farmer’s crop is behind everyone else’s, that farmer may experience significantly more pest pressure.
- Is it socially and culturally acceptable to apply pesticides in the region? If the government restricts use of certain pesticides, farmers should follow regulations and guidelines set by local authorities. If certain pesticide applications are somehow socially unacceptable, farmers should make sure that the community is aware of their IPM plan so that the community can respond in whatever way they feel most comfortable. For example, if a farmer is applying pesticides on crops next to a school, the community may express that this should be done when students are not in school for a certain amount of time. The community may also ask the farmer to put up a hedgerow between his field and the school. Does the community have expressed goals for pest management?
- What management options are feasible, given the available resources? List pest management strategies that are currently used, and record their efficacy (recognizing that this will probably be subjective). What quantities of time, resources, and effort are currently invested in pest management? A lack of resources may limit the number of sustainable and feasible pest management strategies.
Knowledge is an important piece of an IPM strategy. It empowers farmers by increasing awareness of pest traits and helping to shift farmers’ mindsets, so that they proactively focus on prevention of pest problems rather than reactively waiting to see and treat problems that might arrive.
Prevention (Figure 4)
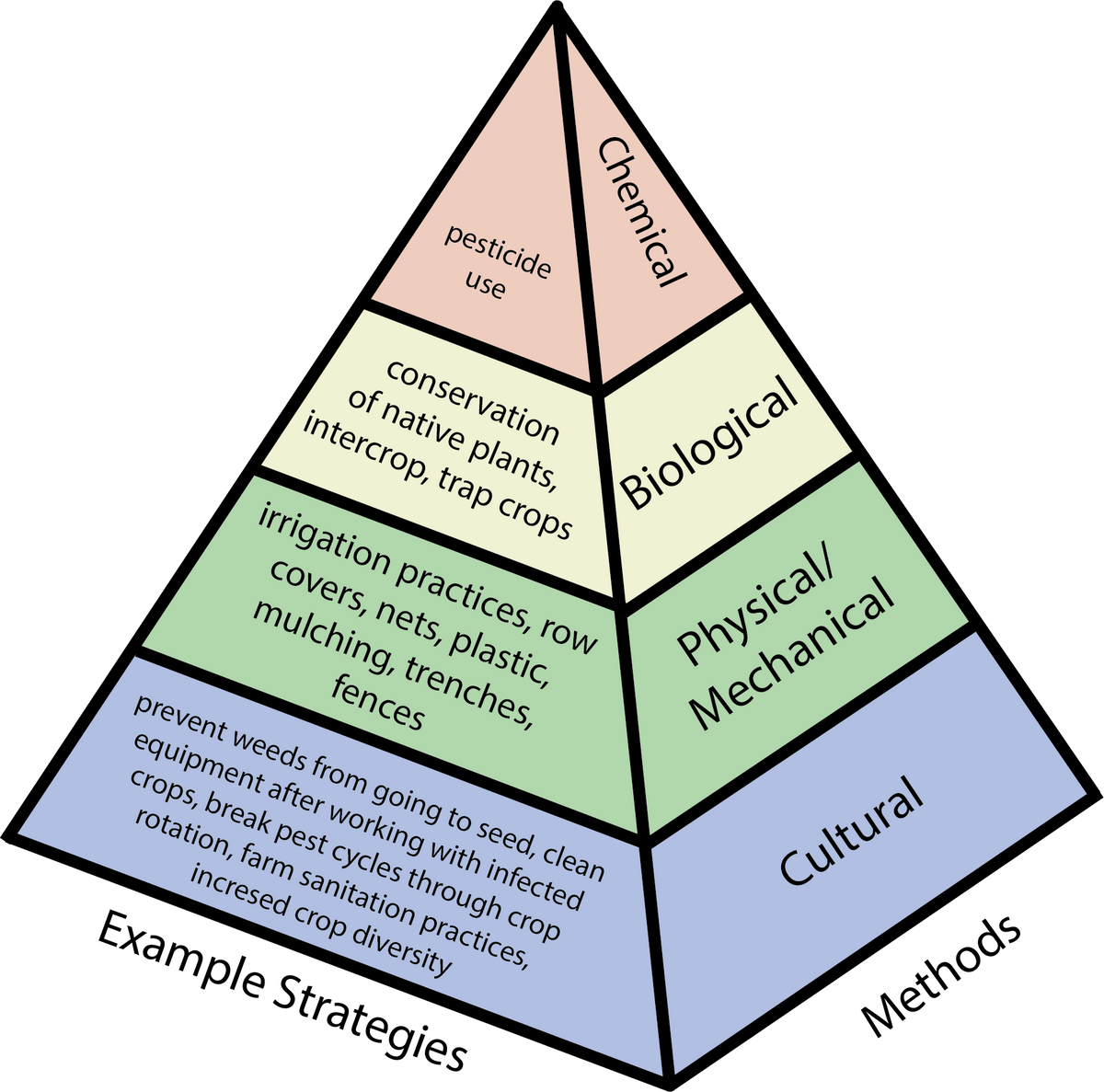
Figure 4. Pest management methods (right) and example strategies (left) for preventative pest control. Source: Stacy Swartz
When the topic of agricultural pest management is mentioned, most people think first about monitoring for pests or intervening to reduce pests: scouting, pest identification, and/or application of pesticides are some specific practices. However, prevention is an often-overlooked key strategy that farmers can use.
Pest prevention (sometimes called pest avoidance) is the deliberate minimization of the potential for pests to be present in the field and is the foundation of IPM. Among all pest control (e.g. biological, physical, cultural, and chemical), some strategies are preventative while others are suppressive. Preventative strategies create unfavorable conditions for pests, for example by limiting pests’ access to water, food sources, or shelter.
Cultural
Select crops or varieties best able to resist pests
Crops differ in their natural defenses against pests, and so do individual crop varieties. Plants actively defend themselves against pests in several ways.
Non-preference plants have some trait (e.g. color, odor, toxicity, or texture) that makes them undesirable to pests. For example, various bean species have trichomes (specific plant hairs) that trap insects or deter them from landing on or laying eggs on leaves. As another example, some plants do not taste good or are toxic, which deters pests from eating them (at least the second time).
Resistant plants respond to pest damage in ways that reduce the amount of damage that the pest can cause. Many resistant cultivars have been developed through crop selection. The International Rice Research Institute has successfully developed various resistant cultivars. Breeding programs for sorghum have led to host-plant resistance for management of sorghum midge, green bug, mites, aphids, and head caterpillars (Sharma, 1993). The International Institute of Tropical Agriculture has developed cassava varieties that are resistant to cassava mosaic disease and cassava brown streak disease (Hahn et al., 1980).
Tolerant plants are more likely to remain relatively healthy and to maintain yield after pests damage them. They can fight off diseases and/or heal after damage has already been done. Crop breeders often select for the trait of tolerance in crops. Tomatoes and cucumbers have varying levels of tolerance to viral infections of tomato yellow leaf curl virus and cucumber mosaic virus, respectively (Pagán and García-Arenal, 2018).
Maintain plant resilience to pests/disease
The health of propagation material contributes to the establishment of a successful crop. Make sure the seed you collect and save is fully developed and mature. Select healthy seed/propagation material that is free of disease, is viable (alive), and has high vigor (is strong). Some diseases spread through infected propagation material such as cuttings. Throughout the growing season, monitor plants for resistance or tolerance, flag those that exhibit higher resistance, and save propagation material from those specific plants.
When plants have access to the resources they need, they can often fight off pests and diseases. When they lack necessary resources, they become weak and are unable to recover as quickly. To minimize these risks, plant on time, at the right spacing, and meet crop water and nutrient demands. This may require split application of inputs (e.g. manure or compost) a few times throughout the growing season, to provide crops with enough nutrients to thrive at different stages of development.
Practice farm sanitation
Pests sometimes remain and multiply on non-crop plants such as weeds that grow among crops. You can help control pest populations by reducing or disrupting pest habitat around the crop. Remove volunteer/weed plants that create habitat for pests, and plants or plant residues that are diseased. Good farm sanitation also includes cleaning equipment after working around infected plants. If you use pruners on infected crops, make sure to sterilize the tool (using heat, isopropyl alcohol, or vinegar) before using it on crops that are not infected, to reduce transfer of disease.
Rotate crops (over time and/or spatially)
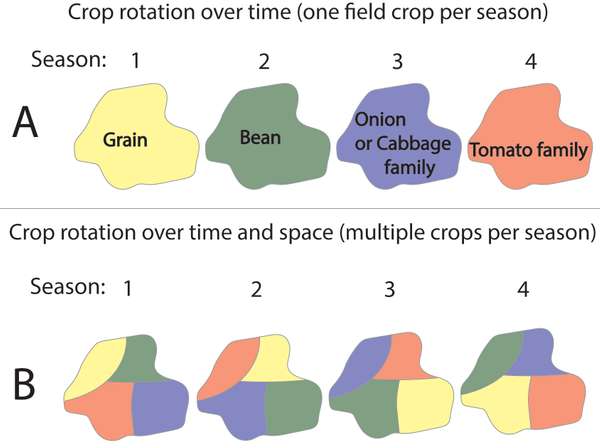
Figure 5. Examples of crop rotations over time (four consecutive seasons/years are illustrated in A) or over space and time (B; colors in B correspond to crop examples in A). Source: Stacy Swartz
Crops in the same family tend to be sensitive to similar pests and diseases. Crop rotation over time and/or space can break the life cycle of a pest by removing the crop host that the pest needs to survive. One option is to rotate a specific field through crops that are sensitive to different pests, over time (Figure 5A). Crops that deter or kill pests can be included in crop rotation schedules; for example, you can rotate crops that are susceptible to nematodes with any number of nematode-suppressing crops (see EDN 75 for some options).
In areas where more than one crop is grown at a time, you can rotate blocks of crops spatially over time (Figure 5B). Switching host crop plants from one location to another will help reduce pest populations over time.
You may want to plant crops with an awareness of when insect pest populations are likely to become a problem. If a particular pest goes dormant for a time (due to cold or dry weather), and you plant ahead of pest emergence, plants can get a head start and gain strength before pests are abundant. However, note that if farmers in your region plant at varying times, some farmers’ crops may experience greater pest pressure because they have planted asynchronously. Be aware of how decisions such as planting time are made at the individual and community levels. Specific preventative cultural examples are found in table 1.
| Strategy | Description | Example |
|---|---|---|
| Break pest cycles through crop rotation | Disrupt the normal pest cycle by crop rotation. | To control for the sweet potato weevil (Cylas formicarius), rotate where in your garden/farm they are planted each year as this is a soil-living insect for its whole life cycle. |
| Break pest cycles through moratoriums | Restrict planting of a crop during a certain time of year. | Each year, when Tomato Yellow Leaf Curl Virus or other white-fly transmitting viruses are most prevalent, no tomatoes are planted on the farm. |
| Farm sanitation | Remove “volunteer” plants | For either sweet potato or tomato situation above, all “volunteer” plants that sprout/grow where and when they are unwanted should be removed to keep pest populations from returning or growing. |
Mechanical
Mechanical preventative measures physically exclude pests from the crop. These include mulches (plastic, paper, or living plants), nets, and row covers (Table 2). Trenches [http://edn.link/jypmaf] and fences [http://edn.link/tn23] are dug or established on the perimeter of the planting area to physically keep pests out of the production area. Mechanical strategies can be effective against one or all stages of a pest’s life cycle. Trenches, for example, are effective against crawling stages of a pest’s life cycle, but do not protect against flying insects. Some mulches can help suppress weeds, but may attract or house pests such as slugs. Mechanical preventative strategies can be labor and resource-intensive so make sure to evaluate the benefits of such strategies before investing in them.
| Strategy | Description | Example |
|---|---|---|
| Netting | Excluding insects from the growing area. | Old mosquito nets that were not soaked in chemicals can be repaired and used to exclude flying insects that are larger than the holes in the net. This is most practical in small plots such as seed production plots. CAUTION: this may also exclude the pollinator for your crop and therefore may need to be removed when the crop flowers. It may also trap birds. |
| Pots/Containers | Using containers with clean soil can minimize impact of soil-borne pathogens or insects. | Growing solanaceous crops (which are susceptible to nematode) such as tomatoes or peppers in pots with soil that has been solarized or from an area high in organic matter can help minimize impact of root-knot nematodes on plant roots and health. |
Biological
Arguably, the most sustainable ways to prevent pest problems over the long term are linked to habitat management. This approach includes designing systems in ways that either divert pests away from crops or increase favorable conditions for pests’ natural enemies. Almost all of the strategies around habitat management will increase a system’s overall plant diversity. Gurr et al. (2003) explain how plant diversity helps with pest management while also keeping inputs low. The authors also describe how the benefits of diversity in agricultural systems go beyond pest management.
Conserving native areas around crops can help prevent pests in several ways. Native plants house beneficial insects that prey on pests or outcompete them for resources. Hedgerows around crops can also visually or physically block the entrance of some pests.
Sometimes a farmer plants a trap crop on the outer perimeter of the plot. A trap crop is a preferred host plant for a pest that also affects a primary crop; by planting it around the perimeter, the pest is led away from the field crop to infest the trap crop instead. On some trap crops, pests cannot complete their life cycle, so farmers do not need to manage the trap crop. In other situations, to break a pest insect’s life cycle, farmers harvest and then destroy trap crops, typically by feeding the plant material to animals or by burning it.
Farmers can intercrop fields with companion plants. Companion plants benefit crops in several different ways. Some deter pests to which cash crops are susceptible or sensitive (see Table 3). Other companion plants attract natural predators of pests.
| Strategy | Description | Example |
|---|---|---|
| Companion plant | Deters pests from crop | Marigolds can reduce a number of pests in various crops including deterring root-knot-nematodes which commonly predate solanaceous crops (for more information about marigolds, see EDN 132 [http://edn.link/marigold]). |
| "Push-Pull" | This system simultaneously incorporates trap crops and companion plants that deter pests. | The International Centre of Insect Physiology and Ecology and the Kenya Agricultural Research Institute system developed a system, described in EDN 116 [http://edn.link/edn-116] that utilizes napier grass or Sudan grass to pull pests out of the crop and desmodium species or molasses grass to push pests out of the crop. |
Chemical
Preventative chemical sprays can be implemented when pests are expected to arrive based on previous season experiences or on a set schedule to keep pests from entering an area or control early-entering pests before populations can increase. This practice utilizes resources that may be limited and may lead to pesticide resistance and so is not highly recommended. Some examples are provided in table 4.
| Strategy | Description | Example |
|---|---|---|
| Apply chemicals to prevent diseases | Leading up to a season when you have noticed diseases issues in the past, applying a chemical pesticide. | Application of copper on fruiting trees such as mango to control for anthracnose and other fungal pathogens. This is most applicable leading up to the rainy season when fungal disease are most prevalent. |
| Apply chemicals to prevent insect pest population buildup. | For highly detrimental pests, some farmers apply chemicals preventatively. | Bacillus thuringienses var. kurstaki for early control of Lepidoptera. If you see adult butterflies or moths around, you may want to take this as an indication to apply preventatively even if you do not have caterpillar damage in your field yet. Make sure with Bt, that you alternate strains (products) so that the pests do not build up resistance. |
After taking precautionary measures specific to your region and implementing practices that prevent pests from entering or multiplying in your production area, you will need to keep a watchful eye on pest populations and intervene before insect pests are likely to cause too much crop damage, or when they already are causing damage.
Observation
When insects are present in low numbers, you may find it difficult to determine whether a particular insect is a crop pest. Pests are often categorized based on their feeding behavior; how quickly they reproduce; whether or not they have natural enemies present; whether they transmit diseases to humans, livestock, or plants; and/or if they contaminate the final food product. Table 5 lists mouthparts and type of metamorphosis of insect orders that include common agricultural pests. The table also gives examples of insect species from each order. The kind of mouthparts and type of metamorphosis are helpful attributes to know about, because they can indicate where an insect pest might be found, the type of damage it might cause, and the best way to treat for it.
| Table 5. Groups, mouthparts, type of metamorphosis, and specific examples of common insect pest orders. | ||||
| Order | Groups | Mouthparts | Metamorphosis | Example |
| Coleoptera | beetles weevils |
chewing | complete | Sweet potato weevil (Cylas formicarius) |
| Diptera | flies | sucking/piercing/lapping | complete | Mediterranean fruit fly (Ceratitis capitata) |
| Hemiptera | true bugs aphids planthoppers |
piercing-sucking | incomplete | Painted leafhoppers (Endria sp.) |
| Lepidoptera | butterflies moths |
chewing (immature stages) | complete | Fall armyworm (Spodoptera frugiperda) and tomato leafminer (Tuta absoluta) |
| Orthoptera | grasshoppers crickets |
chewing | incomplete | Desert locust (Schistocerca gregaria) |
| Thysanoptera | thrips | rasping-sucking | complete (modified) | African thrips (Ceratothripoides brunneus) |

Figure 6. Insect webbing is a substance insects create that sticks leaves together. This photo is of southern beet webworm on amaranth. Source: Annie D.
Metamorphosis describes the way an insect changes from an immature stage to an adult. In incomplete metamorphosis, immature insects look and act very similar to the adults. Insects that undergo complete metamorphosis go through a pupal stage where they change from one form to something completely different (e.g., a caterpillar turns into a butterfly). The immature stages of these insects typically look nothing like the adults, are often found in different locations, and can be much harder to identify. For example, many immature beetles live underground or tunnel into roots, but the adults live on the aboveground portion of the plant. Often an insect is only a pest at one stage of complete metamorphosis; many moths and butterflies do not feed as adults and can even be beneficial pollinators, but the caterpillars (immature/larval stage) can completely defoliate a crop. There will be exceptions to these categories, but understanding the basic ways insects feed and develop is critical for successful pest management.
Insects’ feeding behaviors depend on their mouthparts. Insects with chewing mouthparts remove pieces of leaf or fruit tissue, leaving holes in the plant. Because these insects typically ingest plant tissue, insecticides applied to the surface of the plant may be effective. By contrast, insects with sucking mouthparts do not feed on the outside of the plant, but rather consume sap from within the plant, killing plant cells. In this situation, plant damage can include leaf distortion, yellow stippling, or shriveled grains. Since insects with sucking mouthparts only feed on internal plant juices, insecticides applied to the surface of the plant will not be effective.
Insect feeding damage, webbing (Figure 6), or other insect activity can look similar to many bacterial, fungal (Figure 7), or viral diseases or to plant nutrient deficiency symptoms. Before taking action against an assumed insect pest, verify that the symptoms you observe are not caused by a bacterial, viral, or fungal infection. Insecticides are not effective against these types of infections, and improper use of insecticides wastes farmers’ valuable resources and can kill beneficial insects. For visual comparisons of common diseases and pest damage, view this resource [http://edn.link/photoi].

Figure 7. Leaf symptoms of fungal diseases, such as downy mildew (A) and powdery mildew (B), can look similar to damage from piercing-sucking insects. Downy mildew is restricted by leaf veins, giving it a blocky appearance with white fungal growth on the underside of the leaf. Powdery mildew has small circular discolorations across the whole leaf surface, often forming white spores on either surface of the leaf. Either of these can be confused with damage from piercing-sucking insects (C, lace bug damage on jack bean with lace bug photo inset), which cause pin-point discoloration where they have fed on the underside of the leaf. This can lead to brown necrotic spots that look similar to downy mildew. Source: ECHO Staff
Sampling
The goal of scouting is to estimate the pest population within a field or garden, rather than to count or tally the entire insect population. This sampled estimate allows you to determine whether intervening to control the pest is worth the time, money, and potential negative impacts, or whether you should wait and continue to monitor the pest population.
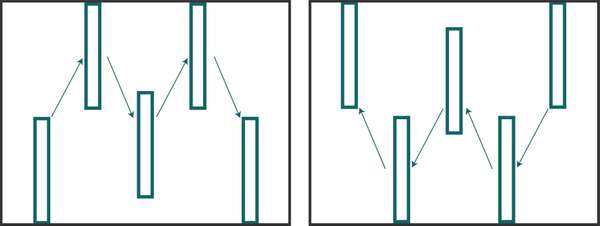
Figure 8. ‘M’ (left) and ‘W’ (right) sample pattern example. Each box would consist of several plants, each of which is inspected for insect presence or activity. Source: Stacy Swartz
You can sample for insects through active behaviors or through passive traps. For both methods, carefully consider where to sample; if you don’t, the results might be meaningless. Insect populations vary in density throughout a field, so be sure to check multiple areas in a random pattern. Many insects will congregate in one area of a field but be present in much lower numbers elsewhere. If you only sample in the heavily infested area, you might think that the pest population is much greater than it actually is. If you sample elsewhere, you might make the opposite mistake of believing that insect levels are very low, when they are not. In general, the best way to actively sample is to walk an ‘M’ or ‘W’ pattern (Figure 8) throughout the field or garden, making 5 to 10 stops to inspect plants for insect presence or activity. For example, when scouting for fall armyworm in maize, it is recommended to make 5 stops in that pattern and inspect 20 consecutive maize plants at each stop. This allows for a random sampling that represents the entire field. When scouting, also pay attention to areas of the field that might have worse damage than others (e.g. field edges often have greater amounts of damage due to points of entry from surrounding areas). In these cases, you could use intervention strategies just in those areas but not the rest of the field. For more information about how to monitor, see EDN 136 [http://edn.link/insectmonitoring] (Liptak and Motis, 2017).

Figure 9. A sweep net made from a pillowcase, thick wire, bamboo, and rubber lashing. Source: Annie D.
Active Sampling
You can accomplish active sampling in a variety of ways. Sweep netting is one way to estimate an insect population throughout an entire field. To construct a basic sweep net from local materials, take a light-colored cloth sack, add a firm wire ring around the opening (30 to 38 cm diameter), and attach it to a stick or pole (Figure 9). Sweep nets work best for low-lying crops such as rice and other small grains, or non-vining beans before they flower and fruit—plants that are tough enough to handle the damage without losing fruit or too much leaf mass. Sweep nets also work best for insects that dislodge easily from the plants.
Beating works well for plants that are too large for a sweep net. To use this technique, tap or shake individual branches over a tarp or sheet and record the number and kind of insects that fall down onto the tarp or sheet. Beating works best in cool temperatures in the early morning, when insects are more likely to fall off the branches than to fly away.
With population indexing, you indirectly measure the pest population by observing signs of insect damage. For example, you could estimate the percent leaf defoliation; amount of insect frass (feces); and/or occurrence of tents, nests, webs, emergence holes, or tunnels in fruit or stems. You could also listen for insect sounds, such as biting or chewing, to help you estimate potential insect damage.
As you scout for evidence of insect pests, be sure to gather and record data (including observations). The information will allow you to monitor changes in the insect population throughout the growing season; it can also help you know when to scout in following seasons. For an example of a scouting record sheet developed by the Canadian Foodgrains Bank, view this document [http://edn.link/faw]. Though originally used for fall armyworm, the resource can be adapted for other pests.
Some pests threaten regional food security. For these, resources may be available to monitor the spread, alert farmers, and create awareness throughout the community and region. Such resources should not replace active scouting in a particular field, but can help you know when to focus on scouting. Examples include the Fall Armyworm Monitoring and Early Warning System (FAMEWS) [http://edn.link/2gp74n] mobile app and Locust Watch [http://edn.link/6mzwhc] (for the desert locust). Both of these are available through the FAO.
Passive Sampling

Figure 10. Farmer using old yellow, plastic container and motor oil (unused) to trap pests. Source: Patrick Trail
Passive sampling typically includes the use of insect traps. Traps range from highly sophisticated and expensive pheromone traps to a container full of soapy water. When making a homemade trap, consider the color of the trap; how the insect will be trapped in or on the trap; and how often you plan to check the trap. As examples, you could use soapy water in colored pans or dishes (often yellow is the most attractive to insects; Figure 10); cards covered with a sticky material and then hung from a tree or placed on a stake in a field; or a pitfall trap made by burying a cup, so insects that walk on the ground fall inside. See research blog post [http://edn.link/j4jfc2] for a comparison of these three types of traps deployed in a sorghum field at the ECHO Global Farm in Florida. Always add a bit of soap to traps containing water, to break the water’s surface tension. Otherwise, insects are small enough that they will stay on the surface of the water and escape. To monitor night-flying pests such as moths, you might consider a light trap. Fruit or other attractants, like meat, can make a trap more effective or can even become the trap itself. For example, in temperate regions, when monitoring for apple maggot (a pest of apples), the trap is sometimes a red plastic ball covered in sticky material and sometimes an actual apple covered in sticky material. Note that these traps are intended to help you determine the pest’s life stage, when they are active, and approximately how many are present. They are not designed to catch enough insects to provide control.
If you are growing a high value crop, you may want to purchase laboratory-prepared pheromone lures or attractants (if available) to help you monitor for specific pests.
Intervention Thresholds
One of the hardest decisions you will need to make as a farmer is to determine when a pest population is high enough that you should intervene to control the population.
The upper damage boundary (also known as the economic injury level; Figure 11) is the point at which the profit lost by the pest damage is higher than the cost it would take to intervene. (If using an insecticide, costs would include materials and the time spent mixing, loading, and spraying.) Ideally, you would never reach this point, because you would intervene earlier.
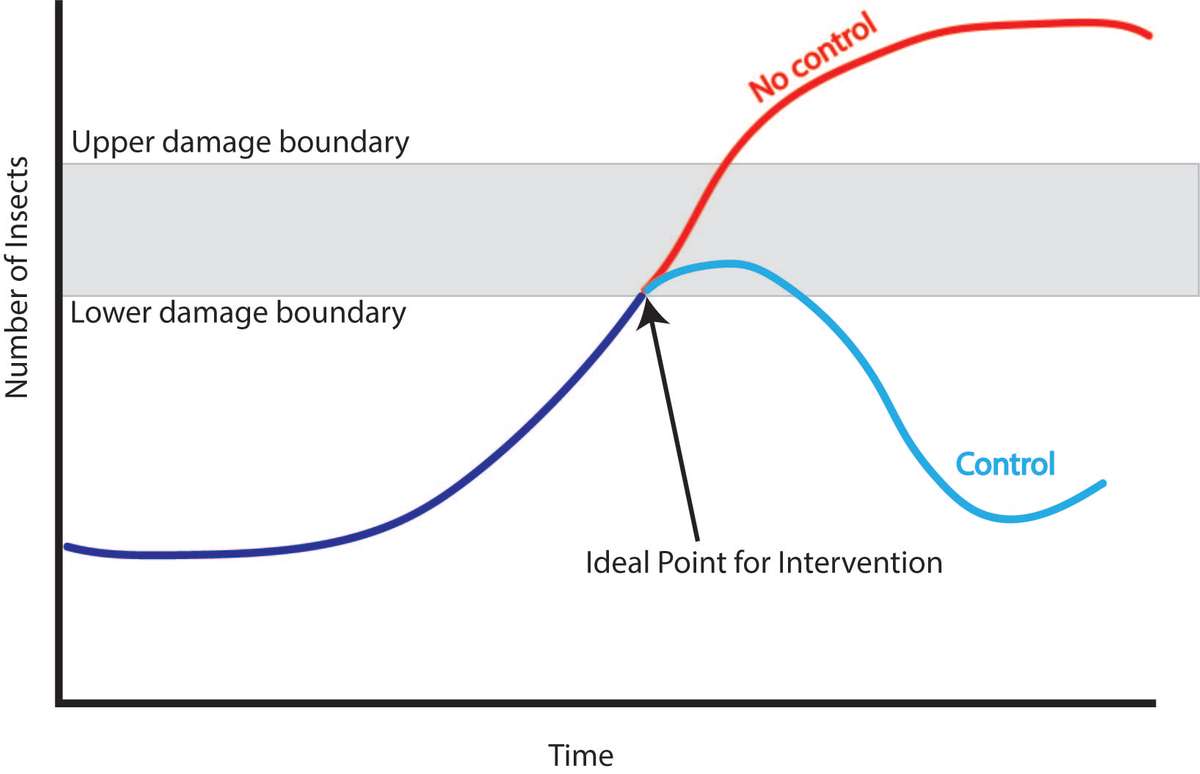
Figure 11. This diagram helps explain intervention timing. The lower damage boundary signifies when intervention should be taken to control the pest population to keep it from reaching the upper damage boundary. If no intervention is made, the pest population will likely go beyond the upper damage boundary. Source: Stacy Swartz
The lower damage boundary (economic threshold) is the action threshold. At this point, you will soon face an economic loss if you do not take action quickly. At the lower damage boundary, you need to make a decision about how to control the pest population to keep it from reaching the upper damage boundary.
The upper and lower boundary levels vary from pest to pest and crop to crop. They depend on a number of factors including crop value, location of insect damage, and crop maturity. Crop value, the most important factor, is based on financial profitability and/or the need for the crop as food for the family. Crops that are worth more have lower damage boundaries. Boundaries also vary based on what type of insect is causing the damage and where on the crop it is feeding. For example, fruit trees can generally tolerate higher numbers of insects feeding on the leaves than insects feeding directly on the fruit. Thus, the damage thresholds for fruit trees will typically be lower for insects feeding on the fruits than leaves. The age of the crop also changes the boundaries, since plants typically tolerate more damage at certain growth stages. For example, newly sprouted seeds or small transplants cannot tolerate as much damage as larger, established plants.
The presence of insects that transmit diseases (vectors) also results in lower damage boundaries. The presence of relatively few of these insects can cause significant harm, well beyond damage from feeding. An example of a vector is Bemisia tabaci, a whitefly that transmits African cassava mosaic virus.
Intervention
A farmer makes critical decisions during a growing season to effectively control pests to preserve the productivity and economic value of a crop. A farmer’s reaction to seeing pests in their crops or grain is to intervene to protect his or her livelihood.
Always prioritize pest control options that fit the local context and utilize locally-available resources. If an intervention requires resourcing from outside the community, you should first consider other alternatives. Try to establish systems that will ensure long-term access and availability of these resources. You may need to:
- support local entrepreneurship,
- create a plan for area-wide management of the pest so that the burden falls on the whole community instead of individual farmers, or
- set up systems for governmental or organizational support.
Additionally, evaluate whether a practice will be culturally appropriate. Seek opinions and feelings of those in the community who might be affected by the pest management plan.
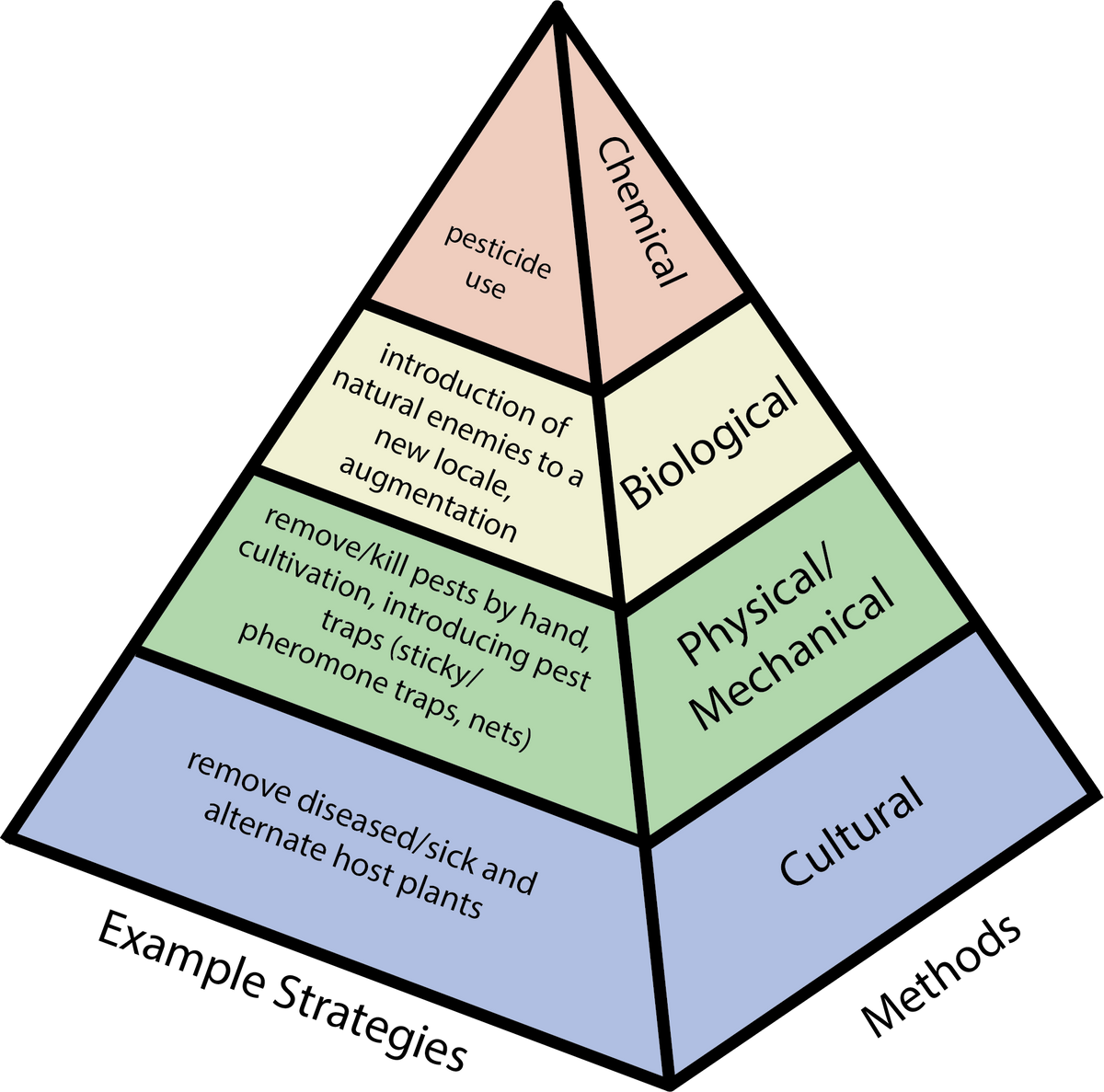
Figure 12. Pest management methods (right) and example strategies (left) for suppressive pest control. Source: Stacy Swartz
When selecting pest control options, utilize interventions that are effective, have the least impact on the surrounding community and environment, and are complementary when integrated. When information for a specific species or crop is not available, you may need to trial potential strategies in a small area to determine which one is most effective. At times, normal seasonal changes are sufficient to keep diseases or pests in check, with no intervention other than adjusting the timing of planting. For example, diseases that flourish under humid conditions may all but disappear during the dry season. Pest control options are discussed under the methods mentioned in figure 12. Suppressive strategies kill or trap pests, reducing existing populations.
Cultural

Figure 13. Potted tomato plant with minimal leaves remaining, especially at the base. Source: Stacy Swartz
Cultural controls modify the environment around plants to make it less favorable for pests. Pests in your main crop(s) may also be harbored/hosted by other species of plants growing in or near your field or garden. Removing or pruning these alternative host plants can help minimize the pest population. If using biological control as well, preserve--as much as possible--trees, shrubs, or weeds near your crop that natural predators use for refuge, food, and nesting. You can also use cultural practices on your main crop (Table 6). Tomato plants, for example, can be managed to minimize bacterial and fungal diseases. Humidity in the leaf canopy is highest around leaves closest to the ground, which is why these water-transmitted diseases typically infect the lower leaves first and then spread upwards to the rest of the canopy. When you remove lower tomato leaves, you reduce humidity near the soil surface by increasing airflow (Figure 13). Doing so early in the season reduces early-season development of diseases.
| Strategy | Description | Example |
|---|---|---|
| Remove diseased parts/individuals | With sterile tools1, prune plant parts (leaves) or whole plants that are heavily diseased or infested. | For Cucurbita (e.g., pumpkins) with excessive downy mildew damage, you can remove leaves that are heavily infested and feed them to animals. Do not mulch with them as the pathogen may still spread. |
| Remove alternative host plants | With sterile tools1, remove alternative host plants that are heavily diseased, infested, or that host vectors. | If you are growing citrus, remove curry leaf tree (Murraya koenigii) or jasmine orange (Murraya paniculata), which are hosts of Asian citrus psyllids that spread citrus greening disease (Gast et al., 2018). |
| Use of efficient irrigation systems | To reduce water-transmitted diseases, irrigate according to plant needs and direct water to the base of plant stems. | Switch from overhead irrigation (which gets leaves wet) to drip irrigation or hand watering at the base of plants. Support plants with stakes or trellising to minimize contact of plant leaves with moisture on the soil surface. |
| 1Tools can be sterilized with isopropyl alcohol, vinegar, bleach, or high heat to kill pathogens that may be on the tool from previous use. | ||
Mechanical

Figure 14. Kaolin clay sprayed on a potted tomato plant. Source: Stacy Swartz

Figure 15. A bucket of soapy water with leaffooted bugs. Source: Annie D.
Mechanical interventions for pest control are generally categorized as passive or active. Passive options include films, dusts, oils, soaps, and traps. Films such as kaolin clay (Figure 14) can deter insects from landing on plants and/or deter feeding behaviors, but such films need to be reapplied as the plant generates new growth. Dusts such as diatomaceous earth can be placed around the base of plants to keep crawling pests from accessing the plant. Dusts can also be placed on the leaves as a feeding deterrent. Oils and soaps that kill pests are physical controls because their effect is short-term and they act physically on the pest by smothering them or breaking down sensitive exterior tissues. Oils and soaps must contact pests and are most effective against soft-bellied insects such as aphids, mealybugs, whiteflies, spider mites, and scales. Repeated application is often required to control a population because oils and soaps are most effective at controlling young individuals. Traps are mainly used to monitor pests but can have applications for reducing pest populations (Table 7). Control of large or dense populations is difficult with traps.
Active mechanical control options include hand-picking and removing pests, vacuums or blowers (pneumatic), and hot water immersion. Picking pests off your plants by hand works well for small areas. Jason Weigner shared that:
This is a great job to get local kids to help with. I was asked in an Ayore village to help with blister beetles that were eating all their hot peppers. There were few enough bushes that we were able to get them under control quickly by turning it into a game with the kids. We made beetle grabbers out of coke bottles with soapy water in the bottom and it was a race to see who could catch the most!
You can pull or squash caterpillars by hand. Beetles, stink bugs, and other larger insects can be put into a bucket of soapy water. The soap breaks the water tension, causing insects to drown in the bucket (Figure 15). Low-tech vacuums suck pests off plants into holding containers that are later dumped into soapy water; this practice is mostly used to collect specimens for pest identification. Hot water immersion effectively kills immature tephritid fruit fly (Diptera: Tephritidae) inside mango fruit (Vincent et al., 2002).
| Stragety | Description | Example |
|---|---|---|
| Picking (by hand) | Pick insects by hand and put them into a container of soapy water or shake them from the plant directly into the container. | Stink bugs (Pentatomidae family) damage tomatoes, rice grain, and much more. Handpick adult stink bugs in the morning, while they are sluggish. This practice can help decrease populations if implemented early (soon after stink bugs are detected). |
| Cultivation | Weeds are removed by scratching the soil surface with a tool. The tool either cuts the shoots from the roots of the weeds or completely buries the weeds. | A stirrup or scuffle hoe is used to cut weeds off just below or at the soil surface. |
| Traps | Traps are often used for monitoring but can also be used to control pest populations in smaller areas by removing specific life stages of the pest or reducing overall numbers. | Sticky traps in nursery or greenhouse settings can help control whitefly populations if installed early when the population is small. These are only effective against adult whitefly. |
| Other mechanical modifications | Altering the environment around plants or seeds can kill certain life stages of pests or whole pest populations. | Consider: Solar heating for cowpea weevil (Callosobruchus maculatus; ECHO Staff, 1992). |
There are also mechanical/physical strategies for managing pests and diseases in stored seed or grain. Polishing grains to remove the outer seed coat with mechanical mills helps remove pests prior to storage. Once in storage, keeping grain in sealed containers excludes pests and humidity; dry conditions prevent the growth of mold. You can also lower or replace oxygen to physically modify the environment in sealed storage containers, killing storage pests or minimizing the damage they cause. Options such as vacuum and replacement of oxygen with carbon dioxide are reviewed by Motis (2020) in EDN 146 [http://edn.link/lowoxygen].
Biological options “add to the answer”
Biological control reduces pests through the management of other living organisms. Biological approaches to pest control are ecologically sound, environmentally safe, and self-perpetuating. Most natural predators are species-specific and, therefore, not a threat to non-target species such as pollinator species [http://edn.link/6ryxxy]. Lastly, biological control brings stability to an agroecosystem over time; this happens as pest populations are reduced, fluctuations in pest populations are moderated, and fewer interventions are needed. Biological control alone is not likely to solve a pest problem. Rather, it supplements other control strategies, supporting long-term effectiveness of an IPM plan.
Natural predators are likely already present and active in your region. You can add a biological component to your IPM strategy by simply enabling these predators to thrive. For ideas on how to attract and sustain natural predators in your garden, read the habitat management information under the ‘Prevention’ section of this article.
You can also re-introduce natural predators that are native to an area but have left the region. This process, called augmentation, is easiest to implement if supporting habitats already exist that can sustain natural predators year-round; without such habitats, the farmer must purchase and release native predators each season. The introduction of non-native predators to control local pests is called classical biological control and is too expensive and risky for most smallholder contexts. Check with local extension services to see what programs exist and what predators are available for distribution and release. Typically, government agencies, educational institutions, or other organizations are responsible for native or exotic predator research and rearing (Table 8).
| Strategy | Description | Example |
|---|---|---|
| Natural predators | A farmer keeps a few host plants for pests, plants that feed adult natural predators, and/or plants that house natural predators to encourage local populations. | Plant a few sunflowers in the off-season, which stink bugs will move to from your crop, encouraging soldier bugs (and other natural predators) to stay in the area. |
| Augmentation | An entomologist goes looking for native natural predators or parasitoids that are no longer in the region and brings them back. | Purchase or apply for parasitic wasps that have been reared by a local university, research center, or government agency. Once obtained, you can release them in your production area. An example of a gardener practicing augmentation was shared by network member Noah Elhardt. 1 |
| Classical biological control | An entomologist looks for non-native potential predators or parasitoids that, when introduced, may control the pest. Researchers rear potential insects, test their propensity to be invasive, and rule out predators that are not good candidates. | Introduction of the predatory wasp (Tamarixia radiata) to help control the Asian citrus psyllid nymphs (Michaud, 2004). This is typically too expensive an endeavor for farmers or even co-ops of farmers to conduct. |
| Microbial disruptors of midgut | Microbes that, after being consumed by a pest, produce protein toxins that create holes in the midgut of the pest. | Some Bacillus sp. are commercially sold in many places around the world and can be applied through a foliar application. Pests then consume the bacteria as they eat crop tissue. |
| Insect-killing fungi | Some fungi are entomopathogenic, meaning they parasitize and complete their life cycles on pest species. | The fungus Ophiocordyceps unilateralis completes its life cycle on only one species of formicine ant (Camponotus leonardi), making a mummy out of the ant’s exoskeleton (Shang et al., 2015). |
Beneficial organisms actively hunt and kill pests for metabolic or reproductive needs. Beneficial predators include many species of ants that consume young caterpillars (Figure 16A), aphids, and other soft-bellied insects. Prionyx wasps hunt and consume grasshoppers (Figure 16B). Beneficial parasites, known as parasitoids, deposit eggs inside a pest, eventually killing the pest (Figure 16C). The most well-known group of parasitoids are the very diverse parasitic wasps. Each species of parasitic wasp lays its eggs inside a highly specific host, which could be a soft-bellied insect such as an aphid or caterpillar or even a hard-shelled beetle. For every crop pest, there is likely at least one parasitoid targeting it. Van Lenteren et al. (2018) outline the use of many diverse species for biological control worldwide.

Figure 16. Carpenter ant (Camponotus sericeus) preying on a caterpillar (A) and predatory wasp (Prionyx sp.) preying on a grasshopper (B). Parasitized hornworm caterpillar. White, fluffy protrusions are parasitoid egg sacs (C). Source: Noah Elhardt (A & B) and Jason Weigner (C)
Biological control is a long-term investment. Parasitic wasps, for example, will not control active caterpillar populations by themselves, but they reduce numbers in future generations. That is why biological control is a vital part of IPM.
Chemical
A brief history of pesticides
Throughout history and across cultures, farmers have used a variety of resources, strategies, and practices to manage in-field insect pressures. Naturally-derived compounds (e.g. sulfur) have been used as pesticides for thousands of years. By contrast, synthetic pesticides have only been produced and used since the 1940s.
Trends in the Global North: The combination of the Industrial Revolution and the Green Revolution in the Global North allowed for the relatively rapid spread and broad use of synthetic pesticides without consideration for human or environmental health. Concerns associated with pesticide use emerged in the 1960s, leading to the establishment of protection agencies. Now, pesticides are largely controlled by governmental regulations that vary among countries, but that generally aim to protect farm workers, pesticide handlers, and the environment.
Trends in the Global South: In the Global South, the access, use, and regulation of synthetic pesticides has varied over time and among regions. Pesticide use in South and Central America has increased over the past three decades, while other regions of the tropics have remained roughly the same (FAOSTAT, 2019; Schreinemachers and Tipraqsa, 2012). Up to 25% of countries in the Global South lack pesticide regulations and (where such regulations exist) their enforcement. This is due to insufficient resources, lack of incentives for enforcement, limited environmental standards, and lack of cohesion between relevant departments (Phung et al., 2012; Schreinemachers and Tipraqsa, 2012; Kegode, 2019). Often farmers are not sufficiently educated about how to properly mix and handle pesticides, dispose of pesticide containers, and use personal protection equipment (PPE). In an effort to unify international standards of pesticide management and to reduce some of the negative effects of regulatory gaps, the FAO and WHO jointly released The International Code of Conduct on Pesticide Management.
Chemical options, as we refer to them in this article, are those that actively work against pests through substances that are toxic to or that repel pests. Chemical control methods include both natural and synthetic pesticides 2. The active ingredient in a pesticide is the portion that is toxic or repellant. The remainder of the pesticide is inert ingredients. It is possible to select natural and/or synthetic insecticides that are “biorational” in the sense that they target specific pests, have minimal impact on the environment, and have low to no toxicity on non-target species.
Mode of action
The mode of action of a chemical intervention describes how a pesticide, whether natural or synthetic, controls the pest. Basic modes of action are summarized in table 9.
| Mode of action | Explanation | Synthetic example(s) | Natural example(s) |
|---|---|---|---|
| Inhibitors | Inhibit pest growth, enzyme synthesis, molting, creation of chitin, or other important metabolic pathways | Organophosphates Methyl bromide Carabamates |
Decaleside (from Decalepis hamiltonii) and Rotenone (found in many plants, including Tephrosia vogelii) |
| Channel blockers | Block channels in neurological pathways or other important metabolic channels (e.g., sodium) | Indoxcarb | Tetrodotoxin (from Taricha granulosa) |
| Modulators | Keep neurological or metabolic systems open, often causing imbalances in one or more senses; a common result is disrupted feeding |
DDT Neonicotinoids Pyrethroids |
Pyrethrin Nicotine Capsaicinoids (in hot peppers) |
| Juvenile hormone mimics | Disrupt and prevent metamorphosis | Analogs of juvenile hormones | Some Echinacea spp. mimic adult hormones (non-lethal distraction) |
| Unknown | The mode of action for some pesticides is still unknown Some pesticides have more than one mode of action |
Azadirachtin (in neem, Azadirachta indica) |
When creating a pest management plan, select pesticides with different modes of action and time their application to vary the mode of action. Diversification of modes of action increases the likelihood of controlling whole pest populations (e.g., all life stages) and prevents pesticide resistance.3 Categories, descriptions, and examples of chemical interventions are given in table 10.
| Strategy | Description | Example |
|---|---|---|
| Natural chemicals | Chemicals extracted from natural sources. These are not always the safest and may still require PPE for application and restrictions on where and when you can use them. | Azadirachtin is extracted from neem seeds and leaves (at a lower concentration) and applied to crops to control a variety of pests, mainly piercing-sucking insects. |
| Synthetic chemicals | Manufactured chemicals. These may be more toxic or less toxic than natural chemicals. | Pyrethroid is a manufactured compound that mimics natural pyrethrins (naturally produced by chrysanthemum flowers). |
| Targeted chemicals | Chemicals specific to a species or group of insects. Such chemicals do not impact organisms outside of a narrow range of target species. | Chlorine containing carabamates such as RynaXypyr® target immature Lepidoptera, but not other insect groups. |
| Broad spectrum chemicals | Chemicals that broadly affect more than just the pest species you are trying to control. | Pyrethrins and pyrethroids kill ants, mosquitoes, moths, flies, fleas, and impact other organisms, including bees and fish. |
| Preventative application | Chemical application before a pest has been identified and sampled. Be cautious in using preventative applications as they may lead to pesticide resistance over time due to frequent exposure of the pest to the chemical. | Application of imidacloprid, a systemic pesticide (absorbed and distributed throughout plant tissue) before any pest presence is identified to ensure control of the Asian citrus psyllid, a vector for citrus greening disease (Gast et al., 2018) |
| Reactionary application | Chemical application after a pest has been identified and sampled. | Application of imidacloprid after a pest the chemical is approved for controlling (mainly piercing-sucking insects) is observed. |
Safety
When using any pesticide, whether natural or synthetic, it is vital to follow correct mixing and loading practices, wear correct PPE, and practice safety measures during application. Pesticide product labels should explain the PPE required for unique settings4 that have distinct PPE recommendations. Product labels should also include any specific health risks corresponding to the product and instructions for how to decontaminate potential spills. Pesticides are sometimes repackaged and sold without labels. If a product does not have a label, try to look up the information with the product and company name, active ingredient, or commonly used name. Refrain from using a pesticide product for which you do not know the name, active ingredient, and potential health risks. Pesticide use without this information is unsafe.
Only use pesticides that are known to be effective on the target pest and apply them at a rate consistent with effective control for that pest. Application rates that vary from recommendations can cause issues with pesticide resistance, environmental contamination, or increased toxicity.
When applying pesticides, there are important factors to be considered:
- Temperature - Avoid spraying pesticides at air temperatures above 30°C, which can burn plant parts. Instead, apply in the early morning or evening when and the temperature is lower, and the sun’s rays are less direct.
- Wind - Do not apply pesticides if there are gusts or consistent wind speeds above 16 kph (10 mph). High wind speed causes your spray to drift into surrounding areas causing damage to non-target organisms, creating a safety issue, and reducing the accuracy of your application.
- Rain - Rain washes off and dilutes many pesticides. Do not apply them if it will rain soon. If it has rained shortly after an application, pay close attention to your crops to determine if reapplication may be necessary.
- Plant physiology - Younger plants are more sensitive to pesticide burns than older plants. Pesticide burns are most likely to occur when the sun’s rays are most direct, and when oils are used. If applying a pesticide with oil in it, spray early in the morning or in the evening. Flowers are more sensitive to pesticides than other plant parts, so try not to apply foliar pesticides when flowers are open.
- Proximity to water bodies - Many pesticides, whether natural or synthetic, negatively impact aquatic or semi-aquatic ecosystems. If you are close to water bodies, observe extra precautions to avoid spray drift, pesticide runoff (can occur if it rains shortly after spraying), or overapplication (can cause leaching into groundwater).
- Effect on target species - It is important to know how a pesticide impacts the target species. It may only kill or repel a specific life stage of the pest and therefore may need to be reapplied later to effectively control a population. For example, eggs are the most challenging life stage to control, and therefore you may need to reapply a pesticide once eggs of the previous generation have hatched.
- Effect on non-target species - It is essential to understand how a pesticide may affect non-target species. This consideration is important if you are hoping to create long-term health and balance in your agroecosystem. If there are harmful effects on pollinator species, biological control agents, or other organisms that are desirable in your agroecosystem, reconsider your use of the product; explore other alternatives or apply to plants before they flower (to avoid harming pollinators).
- Timing and frequency of application - Many natural pesticides are less effective than synthetic pesticides. Therefore, some natural pesticides need to be applied when pest populations are low and be applied frequently to ensure the pest population stays within acceptable levels.
If you apply a pesticide that may harm people and animals, communicate with local community members to ensure that everyone knows when and how long they need to stay out of the area. Commercial pesticides have published restricted entry intervals (REIs) 5 that indicate when it is safe to re-enter the sprayed area.
Integration of interventions
Cultural, mechanical, biological, and chemical intervention options should be leveraged when they are most appropriate for your context, crop needs, and level of damage. Select practices from each category that fit your needs, resources, and comfort. It is unlikely that any one practice can effectively control pests while maintaining long-term sustainability. Therefore, it is best to integrate your pest management approach with a variety of strategies that trap, repel, and reduce pests in unique ways (Figure 17). Damage boundaries also vary based on the control measures available. Jason Weigner shared a caution about suppressing pests to levels that will not sustain natural predator species:
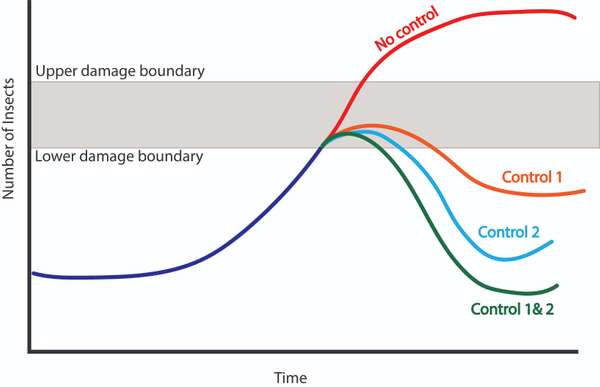
Figure 17. This diagram shows how different types of control strategies could impact a pest population. Control 1 doesn’t reduce the population as much as control 2, but last a bit longer in its effectiveness. Combining both controls may have an even greater effect in reducing pest population. Source: Stacy Swartz
[Reducing] the pest population too low could lead to the predator dying off or moving on [to another area], which then leads to worse pest spikes in the future.
An example of an IPM approach for dealing with sugarcane white grub in Bali, is described in Asia Note 42 [http://edn.link/qt6hxz]. Start by considering practices at the base of the pyramids (Figures 4 &12) and work your way up if possible. If there is too much damage already done, this may not be possible, and you may have to start with stronger suppressive steps. Next season, try to invest time earlier in the season to implement cultural, mechanical, or biological options.
Evaluation and Assessment
After attempting to prevent or control a pest, farmers should monitor pest activity to accurately evaluate the effectiveness of their interventions and overall management approach. Monitoring pests and evaluating the effectiveness of a preventative strategy may require time and patience as many preventive strategies take weeks, months, or even years of implementation. Monitor preventative strategies intensively at the beginning of a cropping season to determine if the strategy keeps pest populations from building up or entering the field. To evaluate the efficacy of a suppressive strategy meant to control existing pests, monitor pest populations in the short-term—within a few days of the intervention to ensure crop protection. If utilizing a chemical intervention to control a pest, begin monitoring pests only after it is safe to reenter application areas. REIs should be listed on product labels or found by researching the pesticide’s active ingredient or common name. Table 11 lists some examples of different control strategies and some estimated timelines for monitoring.
| Strategy | Type of control | Category of control | Monitor timeline |
|---|---|---|---|
| Trenches (e.g. beetle control) | preventative | cultural | Monitor within the first season, especially the beginning of the pests’ life cycle in the region. |
| Attracting beneficial insects | preventative | biological | Monitor over several years, including during the off-season when crops are not growing. |
| Systemic* pesticide spray | suppressive | chemical | Monitor in the following 3-10 days after spraying. Systemic pesticides can take several days to travel to all parts of the plant after uptake to be effective. |
| Soap and oil spray | suppressive | mechanical/physical | Monitor after the soap and oil dry (within 2 days). Soap and oil act on the exterior of the pest- suffocating it or breaking down outer tissues. The spray is only effective as long as it stays wet and makes contact with the pests. |
| * Systemic pesticides are those that a plant absorbs then distributes throughout plant tissues to reach all plant parts. | |||
Monitoring pest levels can be done with the same techniques as overviewed in the ‘observation’ section of this article (e.g., various traps, nets, etc.). Pest activity may differ depending on what interventions are used. Some interventions are toxic, causing the death of a pest at one or more life stages. Others are deterrents, which cause pests to leave the crop. Still others cause changes in pest behavior, such as the inability to molt or feed, and take more time to have a lethal effect than toxic interventions. Therefore, it is helpful to know how a strategy affects a pest when monitoring.
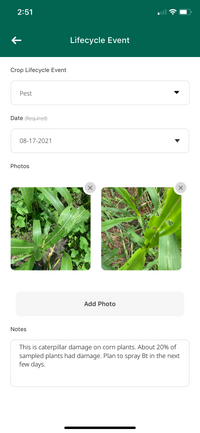
Figure 18. A screenshot of the ECHOcommunity app after entering pest management observations into the life cycle event feature. Source: ECHOcommunity app
When monitoring, record observations about how well pest management interventions are working, and what factors might be influencing their effectiveness.
- Which life stage(s) of targeted pests are being impacted?
- How lethal/deterring is the intervention for each life stage?
- How long does it take to affect the pest?
- Is anything unique about the weather or environment that may impact the effectiveness of a strategy?
These notes are helpful when assessing which interventions to include or exclude from an IPM plan. The ECHOcommunity app (Figure 18) allows the user to track and record information under the “My Plant Records” feature. Select ‘Add a Lifecycle Event’ -> ‘Pest’ and record information in the notes section. Useful notes to record are the management strategy, monitoring notes (both before and after implementing the strategy), and other observations. The feature also allows photos to be added to the entry.
Monitoring is essential not only for evaluating the effectiveness of an intervention but also for the continued protection of a crop. If a strategy does not control a pest population, repeat or use another strategy.
Evaluate strategies
Many factors impact how effectively a pest management strategy works. Rain shortly after a foliar application may limit how much of a product remains on the plant to control the pest. Air temperature and sunlight intensity can impact how quickly a pesticide (natural or synthetic) breaks down and, therefore, how long a pest is exposed to it. Pest exposure to a pesticide is also influenced by how evenly it is sprayed, with what size of droplets, and what surface (e.g., undersides, top of the leaves, or entire canopy) of the leaf was sprayed.
A farmer’s considerations are vital to a successful and meaningful IPM plan. A sustainable, long-term investment in pest mitigation is only as good as it is acceptable to a farmer or farming community. Therefore, it must be the farmer’s assessment that determines the value of a strategy. Acceptable damage to a crop is dependent on a number of factors, many of which are unique to a specific farmer’s context. Factors farmers should consider in evaluating the merit of a strategy are overviewed in table 12.
| Factor | Explanation |
|---|---|
| Intended use of crop | Pest damage on a fodder crop such as napier grass (Pennisetum purpureum) is generally more acceptable than pest damage on an ornamental crop such as hibiscus (Hibiscus spp.). |
| Maturity of the crop | Plants are more susceptible to damage when young, so interventions are often more beneficial when applied earlier rather than later in a crop's life cycle. |
| Resource availability | If a farmer must travel a far distance or cannot find the materials needed for a strategy, it will lessen the value of the strategy. |
| Resource accessibility | If a farmer cannot travel to get materials due to infrastructure limitations or weather conditions, or cannot afford materials, it will lessen the value of the strategy. |
| Information availability | If a farmer cannot find information about a pesticide or strategy, it will lessen the value of the strategy. |
| Information accessibility | If a farmer is illiterate and the information available is all written or if strategy instructions are not in their native language, it will lessen the value of the strategy. |
| Cost of strategy | The ratio of cost to effectiveness of a strategy must be economically favorable for the farmer. |
| Time constraints | If a farmer does not have time to spray a pesticide multiple times, a strategy that requires repetition would be less valuable. |
| Labor constraints | If a farmer does not have enough energy or laborers to implement a strategy (trenches), it will lessen the value of the strategy. |
| Toxicity | If a strategy is highly toxic (high lethal rate) to the pest, a farmer may value it more. If a strategy is highly toxic to non-target species, such as bees, a farmer may value it less. |
| Environmental impact(s) | If a strategy negatively impacts the environment, a farmer may value it less. |
| Societal opinions | If the community does not accept, value, or feel safe with a strategy, a farmer may value it less. |
| Traditional practices | If a strategy goes against traditional practices, it may be difficult for a farmer to switch and go against cultural norms. |
Development workers, extension agents, and other practitioners can help guide farmers by asking questions about their satisfaction with a strategy they used or simply monitoring the crop alongside the farmer and asking them to share about the successes and challenges of the cropping season.
Make Goal-Oriented Adjustments to IPM Plan
Setting goals can come at any point in the IPM decision-making process, but a good time to reevaluate them is after intervening to control a pest population. As much as possible, consider sustainability as the overarching goal for pest management plans. A plan's economic, social, and environmental sustainability are all important for the continued long-term control of agricultural pests. Having and readdressing a set of goals can solidify a pest management plan around core ideas that are important to a farmer or farming community.
An example set of goals is:
- Mitigation of crop loss (no less than a minimum number of kg/acre)
- Long-term reduction of pest presence and pressure
- Low impacts on the environment
- Acceptability by the surrounding community
Now it is time to close the loop of IPM by assessing which strategies to use for future seasons, including modifications learned or adopted as well as lessons learned from previous seasons. IPM is a continual process (Figure 1) that should improve over time as you gain experience, observe pests, implement strategies to control pest populations, and evaluate the decisions you have made.
By learning, observing, and acting, you can evaluate outcomes and determine if pest management decisions are in line with your goals. This process is strategy assessment.
One approach to assessment is to list strategies under each goal that they address in order of effectiveness. More frequently listed and higher-ranked strategies should be kept/added to a pest management plan. Delete strategies that you will no longer implement, noting why they will be discontinued. For example:
- Rotate through different modes of action for stink bug chemical sprays.
- Establish white-flowering plants with nectaries for beneficial predators of stink bugs.
- Communicate with neighboring farmers about the timing of interventions to control stink bugs so that we are more synchronous in our control efforts.
Companion plant sunflowers to attract stink bugs away from main crop:too labor-intensive and didn't seem to make that big of a difference for how much labor it required. May not have timed the planting correctly to effectively trap stink bugs.
Potential Constraints
Smallholder farmers face several hurdles that make it difficult for them to enact an IPM plan. Some farmers who have fields that are far from their home may only travel to the field for important management activities such as planting, weeding, and harvesting. This limited time in the field is sometimes insufficient to catch pest populations at levels that can be controlled in a timely and effective manner. Farmers may be unable to scout fields often enough without using valuable resources (e.g., time and travel expense).
Generally speaking, little information exists on the topic of pest management using locally available resources in tropical environments. Temperature and crop species (plant host of the pest) affect the life stage length of many pests (e.g. Nava-Camberos, et al. 2001 6). A shorter life cycle means an insect pest species can go through multiple generations and can multiply rapidly. However, little is known about how in-field tropical conditions affect pest life cycles. Also, little research has been done to better understand alternate tropical host species of many of the major agricultural pests and diseases.
Many regionally-practiced intervention strategies for controlling pests exist, but information is limited on which practices are effective against specific pests and under what conditions. Tesfaye and Gautam (2003) reviewed 26 traditional pest management practices used in India and Ethiopia, many of which need validation or revisiting. There is a great need for additional evaluation of locally-practiced pest control strategies and the adaptation of effective pest control to regional contexts.
Network member Jason Weigner shared:
Anyone working with local farmers should spend time asking about local pest management techniques. Odds are you'll find things that don't really work, but you may also find some real gems. I have been researching and experimenting with ways to control leafcutters for years. Unfortunately, many things I've come up with are expensive, time consuming, or involve chemicals. Then one day I was walking with a Bolivian guy I work with looking at trees that had been defoliated by leaf cutter ants yet again and he casually said "We need to get some cotton.” The moment he told me about wrapping the trunks with cotton it made total sense. They get tangled in the cotton fibers, so they go hunting elsewhere. Such a simple and inexpensive solution found locally.
Perceptions of different control strategies vary from one region to the next. Where synthetic insecticides are available and accepted, farmers are not always informed about the proper mixing and handling of pesticides, disposal of pesticide containers, and use of PPE. Moreover, pesticide handling and regulation efforts are not always consistent or enforced. Constraints such as these will shape how farmers plan their pest management.
Conclusion
Smallholder farmers in the tropics face increasingly overwhelming hurdles. Population growth, a volatile global climate, and the need for long-term productivity make sustainable food production challenging. Integrated pest management is a platform to equip farmers with diverse decision-making skills. When farmers focus on knowledge and preventative measures, they begin to feel in control of their production, and their mindset about pest management changes from reactionary to preparatory.
When it comes to insect pest management, look for local sources of expertise. Regional agricultural universities, government agencies, or agricultural organizations may host local extension services. Trained professionals may be able to help with pest identification, early detection, and scouting practices proven to be effective in the region. Seek out these professionals’ local expertise and knowledge before taking more general approaches, such as those outlined in this article. If your area lacks such experts, you may need to depend on information that have been utilized for adjacent or similar situations.
Farmers know and understand their fields and crops best because they are present in their fields the most and know the seasonal history of regional crop production. Farmer’s inherent creativity to solve pest problems has the potential to bring IPM strategies forward and should be encouraged. Look for opportunities to glean insights from farmers on how to effectively control pests and enhance the overall balance of agroecosystems.
Further Reading
To learn more about natural pest control options, explore oisat.org [http://edn.link/rxj2dy], where you can navigate resources by pest, crop, or control strategy.
The USDA has a section on “Problem Prevention and Holistic Pest Management” specific to tropical nurseries starting on page 273 of their Tropical Nursery Manual: A Guide to Starting and Operating a Nursery for Native and Traditional Plants [http://edn.link/2mtf7j].
For a facilitator's guide on insect identification and monitoring, see the Canadian Foodgrains Bank's training module on Insect Identification and Monitoring [http://edn.link/9r7z6f].
Rapisarda and Cocuzza’s Integrated Pest Management in Tropical Regions [http://edn.link/7x933r] book published by the Centre for Agriculture and Bioscience International is a valuable resource. The book goes deep into practices, integration of control options, and specific constraints in tropical conditions, including the unique impacts of climate change on pest management.
ORGANICAFRICA has training manuals in English, French, and Swahili that cover organic and sustainable agricultural practices in a 10-module series. Module 4 goes in-depth into pest, disease, and weed management in specific. This module also contains graphic posters and booklets for instructional use.
The FAO has regionally-specific resources related to IPM programs in the Near East and West Africa. These resources include training manuals (in local trade languages), stories from farmers, and more.
References
Chakraborty, S. and A.C. Newton. 2011. Climate change, plant diseases and food security: an overview. Plant Pathology 60:2-14.
ECHO Staff 1992. Short term heating kills cowpea weevils. ECHO Development Notes no. 37.
FAO. 2017. Fall armyworm spreads to East Africa. FAO in emergencies. Accessed September 9 2019 http://www.fao.org/emergencies/fao-in-action/stories/stories-detail/en/c/882822/
FAOSTAT. (Accessed September 19, 2019). Search terms: Agri-Environmental Indicators, Pesticides (Total), and use per area of cropland. http://www.fao.org/faostat/
Gast, T. and T. Watkins, summarized by Stacy Reader 2018. Yellow shoot, green fruit: citrus greening disease. ECHO Development Notes no. 138.
Gurr, G.M., S.D. Wratten, and J.M. Luna. 2003. Multi-function agricultural biodiversity: pest management and other benefits. Basic and Applied Ecology 4(2):107-116.
Hahn, S.K., E.R. Terry, and K. Leuschner. 1980. Breeding cassava for resistance to cassava mosaic disease. Euphytica 29(3):673-683.
Kegode, G.O. 2019. Synthetic Pesticides in Africa: the Good, the Bad, and the Ugly. Feed the Future. Accessed September 11 2019 https://www.agrilinks.org/post/synthetic-pesticides-africa-good-bad-and-ugly
Liptak, C. and T. Motis. 2017. Crop monitoring for early detection of insect pests. ECHO Development Notes no. 136.
Manandhar, A., P. Milindi, and A. Shah. 2018. An Overview of the Post-Harvest Grain Storage Practices of Smallholder Farmers in Developing Countries. Agriculture 8(4):57.
Michaud, J.P. 2004. "Natural mortality of Asian citrus psyllid (Homoptera: Psyllidae) in central Florida." Biological Control 29: 260-269. doi:10.1016/s1049-9644(03)00161-0.
Motis, T. 2020. Low oxygen methods for insect control in seeds. ECHO Development Notes no. 146.
Nava-Camberos, U., D.G. Riley, M.K. Harris. 2001. Temperature and host plant effects on development, survival, and fecundity of Bemisia argentifolii (Homoptera: Aleyrodidae). Environmental Entomology 30(1):55-63.
Omkar. 2018. Pests and Their Management. In: Omkar (Ed.) Pests and Their Management. Springer, Singapore.
Pagán, I., and F. García-Arenal. 2018. Tolerance to plant pathogens: theory and experimental evidence. International Journal of Molecular Sciences 19(3):810.
Peshin, R., R.S. Bandral, W.J. Zhang, L. Wilson, and A.K. Dhawan. 2009. Integrated Pest Management: A Global Overview of History, Programs and Adoption. In: Integrated Pest Management: Innovation-Development Process; R. Peshin and A.K. Dhawan, Eds. Springer, Dordrecht.
Phung, D.T., D. Connell, G. Miller, S. Rutherford, and C. Chu. 2012. Pesticide regulations and farm worker safety: the need to improve pesticide regulations in Viet Nam. Bulletin of the World Health Organization 90(6):468-473.
Sallam, M.N. 1999. Insect Damage: Damage on Post-harvest. FAO and International Centre of Insect Physiology and Ecology (ICIPE).
Schreinemachers, P, and P. Tipraqsa. 2012. Agricultural pesticides and land use intensification in high, middle and low income countries. Food Policy 37(6):616-626.
Shang, Y., P. Feng, and C. Wang. 2015. Fungi that infects insects: altering host behavior and beyond. PLoS Pathog. 11(8).
Sharma, H.C. 1993. Host-plant resistance to insects in sorghum and its role in integrated pest management. Crop Protection 12(1):11-34.
Sisay, B., J. Simiyu, E. Mendesil, P. Likhayo, G. Ayalew, S. Mohamed, S. Subramanian, and T. Tefera. 2019. Fall armyworm, Spodoptera frugiperda infestations in East Africa: assessment of damage and parasitism. Insects 10,195.
Stork, N.E. 2018. How Many Species of Insects and Other Terrestrial Arthropods are there on Earth? Annual Review of Entomology 63:31-45.
Tesfaye, A., and R.D. Gautam. 2003. Traditional pest management practices and lesser exploited natural products in Ethiopia and India: Appraisal and revalidation. Indian Journal of Traditional Knowledge 2(2):189-201.
Van Lenteren, J.C., K. Bolckmans, J. Köhl, W.J. Ravensberg, and A. Urbaneja. 2018. Biological control using invertebrates and microorganisms: plenty of new opportunities. BioControl 63:39-59.
Vincent, C., G. Hallman, B. Panneton, and F. Fleurat-Lessard. 2002. Management of agricultural insects with physical control methods. Annu. Rev. Entomol 45:261-281.
Cite as:
ECHO Staff. 2022. Integrated Pest Management. ECHO Technical Note no. 98